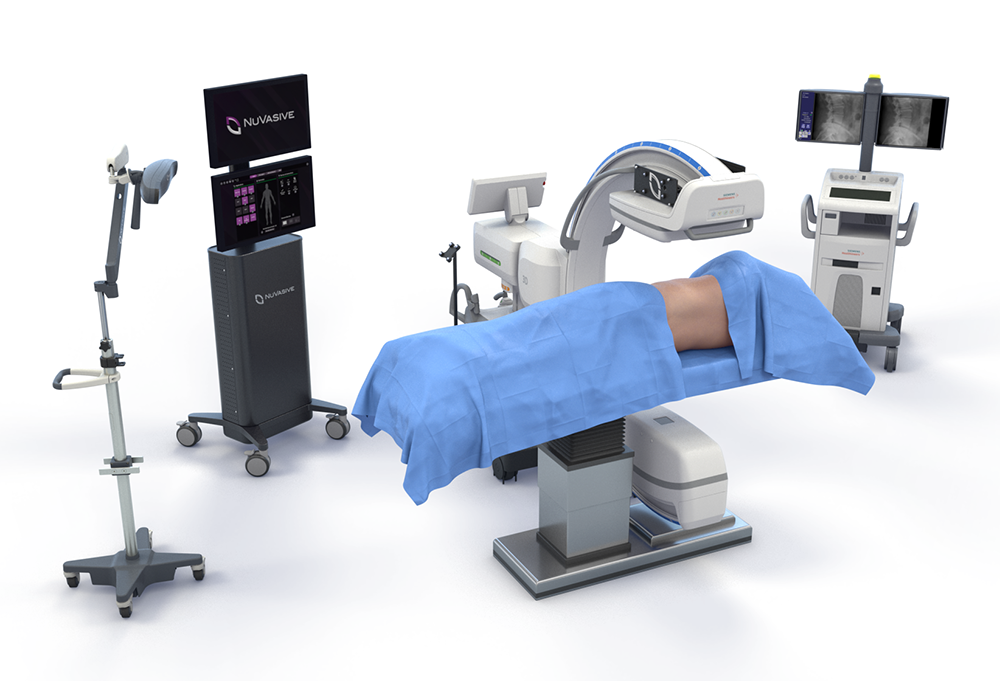
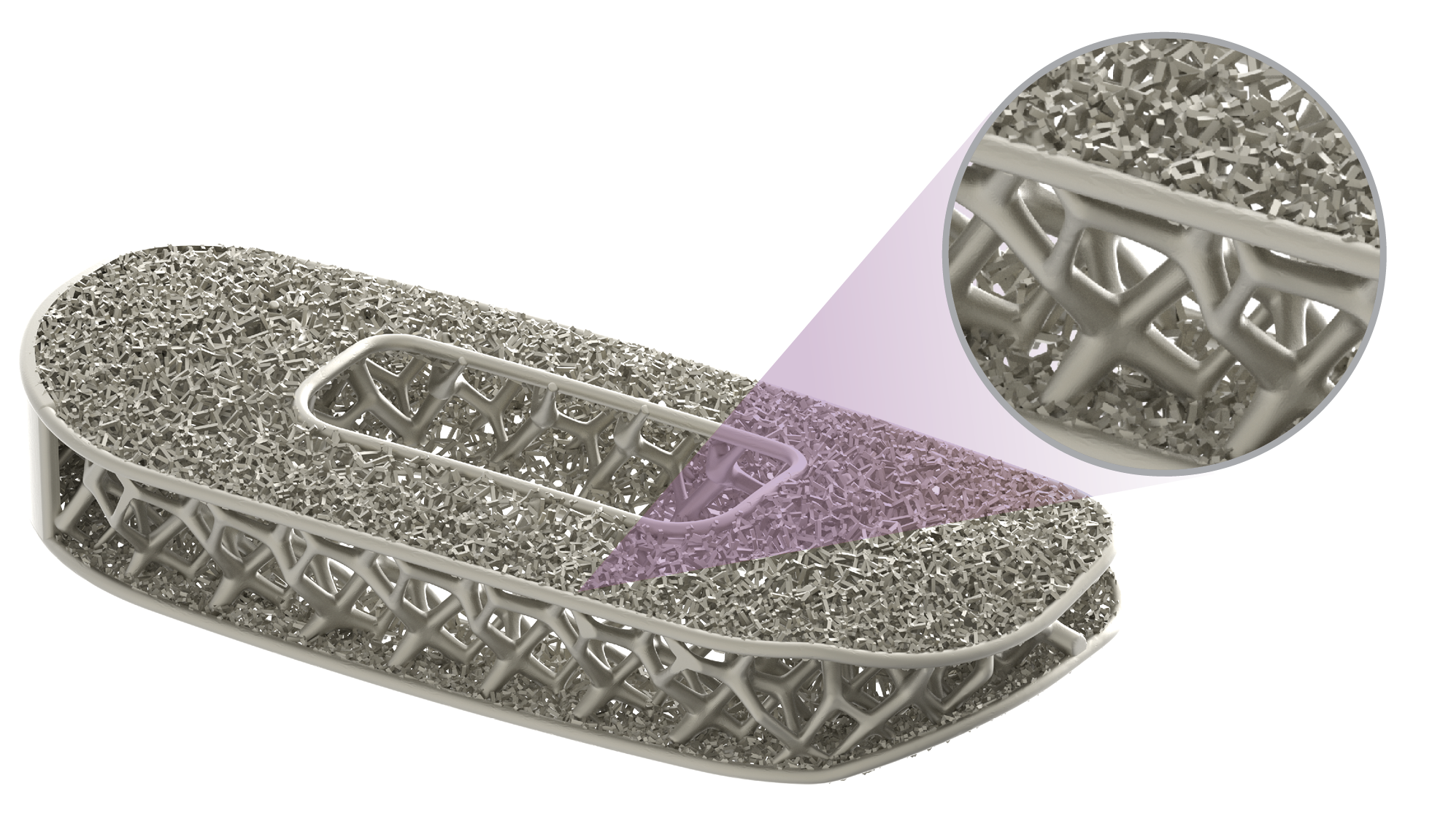
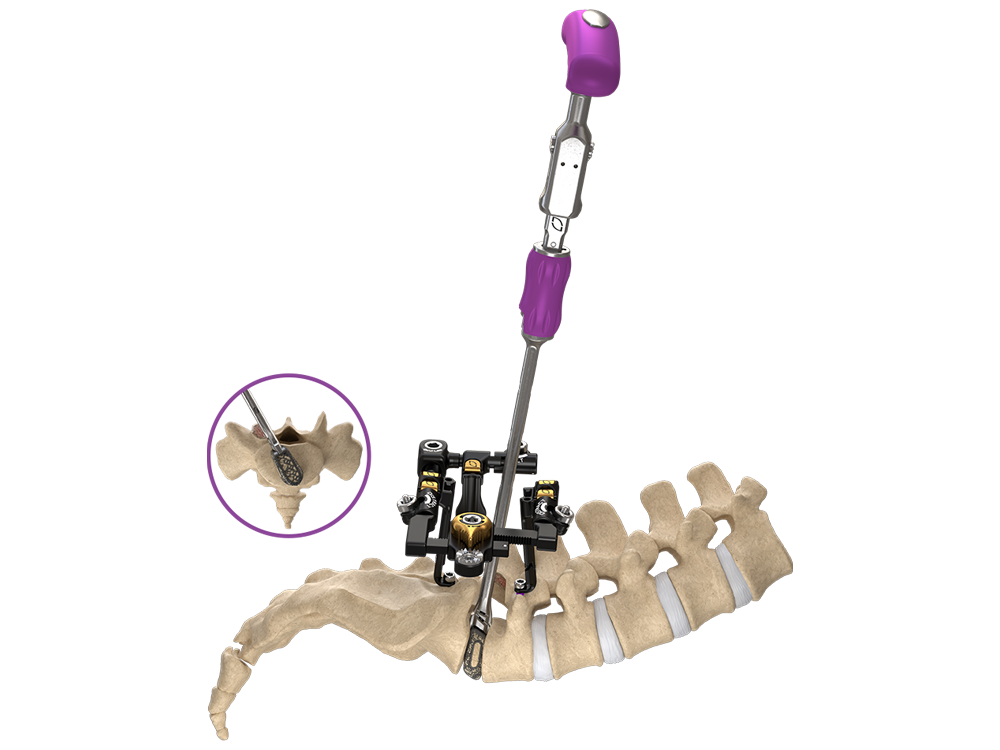
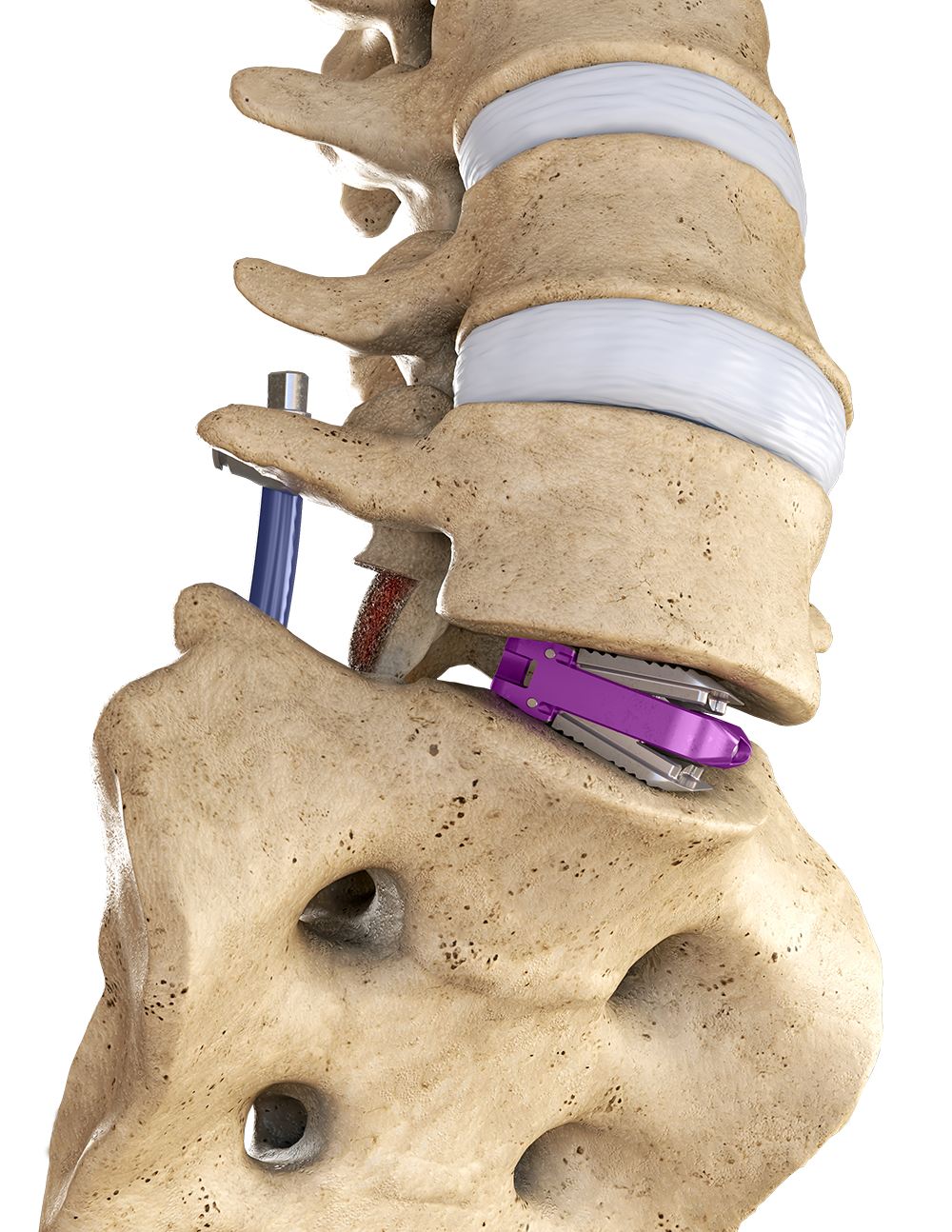
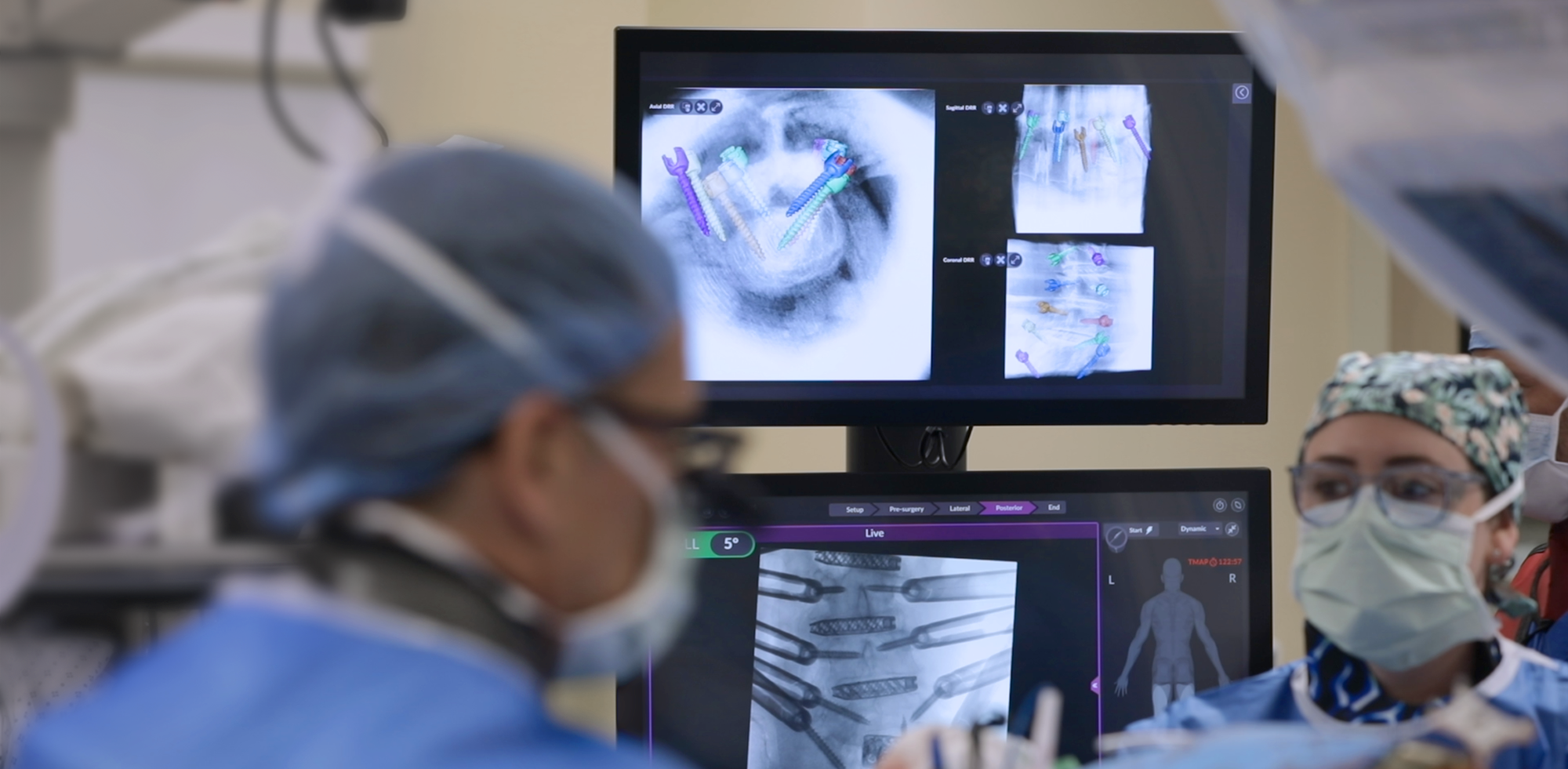
Uniting to advance patient care
Globus Medical and NuVasive have officially joined forces. We’re committed to providing innovative technologies and industry-leading clinical support to help surgeons and healthcare providers deliver better care around the globe. We now have one of the most comprehensive offerings of musculoskeletal procedural solutions and enabling technologies to impact the care continuum. Our employees are relentlessly focused on changing patient lives.