SAN DIEGO – October 30, 2018 – NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced financial results for the quarter ended September 30, 2018.
Third Quarter 2018 Highlights
- Revenue increased 9.8% to $271.3 million, or 10.2% on a constant currency basis;
- GAAP operating profit margin of 6.6%; Non-GAAP operating profit margin of 15.6%;
- GAAP diluted earnings per share of $0.30; Non-GAAP diluted earnings per share of $0.56; and
- Company updates full-year 2018 guidance.
“Our third quarter results reflect accelerated year-over-year revenue growth of nearly 10%, supported by strong performances in both spinal hardware and surgical support business lines with overall U.S. case volumes up more than 7%,” said Gregory T. Lucier, chairman and chief executive officer of NuVasive. “With the sense the overall U.S. spine market is trending healthier, we made strategic investments this quarter on the heels of this momentum in key R&D initiatives, additions to our commercial sales force and infrastructure upgrades to improve set fulfillment—all to support a strong start to 2019 and beyond.”
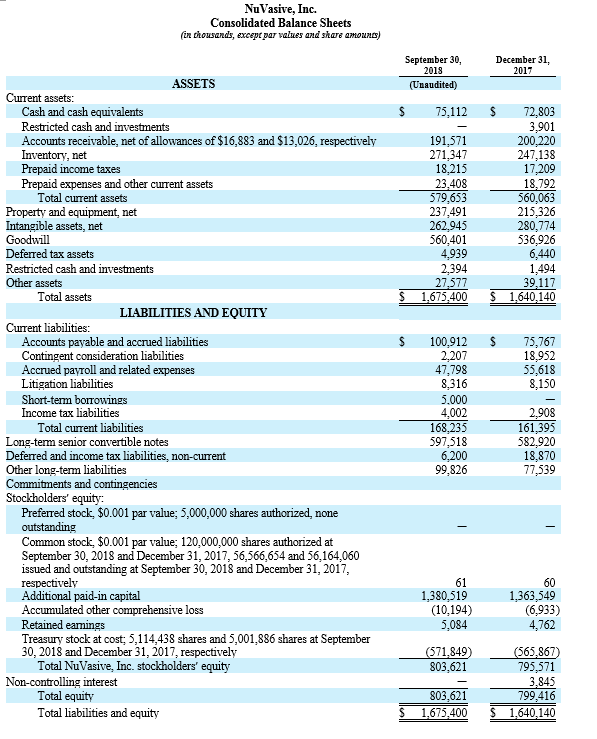
The Company’s financial results reflect continued improvement of its in-sourcing efforts at the West Carrollton, Ohio manufacturing facility, and the Company reiterated expectations that the facility will drive an additional 130 to 150 basis points in operating margins in 2019.
Lucier commented, “We made solid progress with our in-source manufacturing initiatives by bringing in additional SKUs during the third quarter with throughput ramping to higher volumes. This strategic investment is on track and will become a business advantage, both to drive a competitive cost position and to control the quality required to produce evermore complex implants.”
NuVasive also recently made several key technology introductions and partnership announcements, including the unveiling of the NuVasive PulseTM surgical automation platform, Spine Precision Partnership with Siemens Healthineers and signing of a strategic partnership with Biedermann Technologies to further enhance NuVasive’s best-in-class complex spine deformity technologies. The Company also launched several new products to further reinvigorate its Biologics business line, which continues to recover at a faster-than-expected pace. The Company now anticipates its Biologic business will return to growth in the fourth quarter 2018.
A full reconciliation of GAAP to non-GAAP measures can be found in the tables of this news release.
Third Quarter 2018 Results
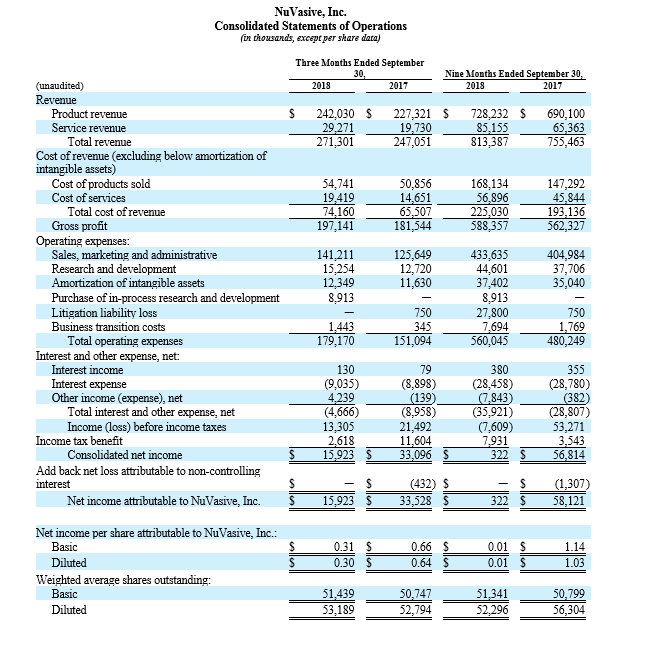
NuVasive reported third quarter 2018 total revenue of $271.3 million, a 9.8% increase compared to $247.1 million for the third quarter 2017. On a constant currency basis, third quarter 2018 total revenue increased 10.2% compared to the same period last year.
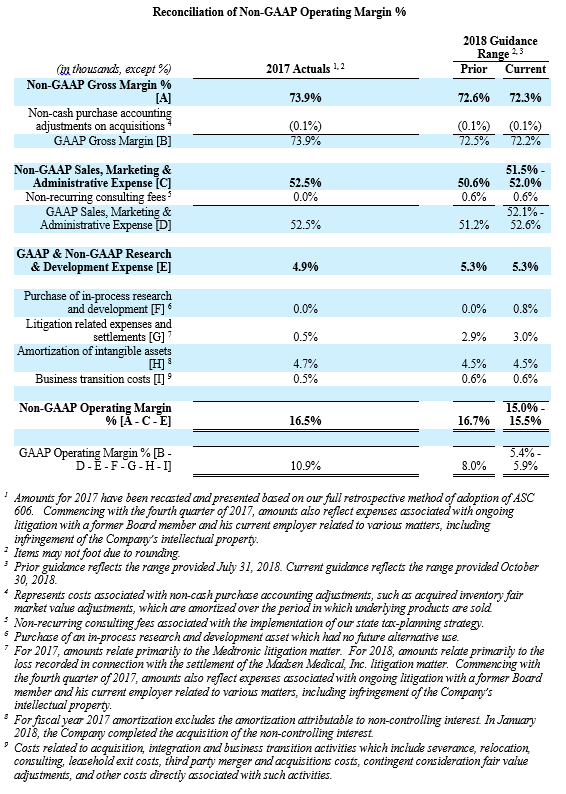
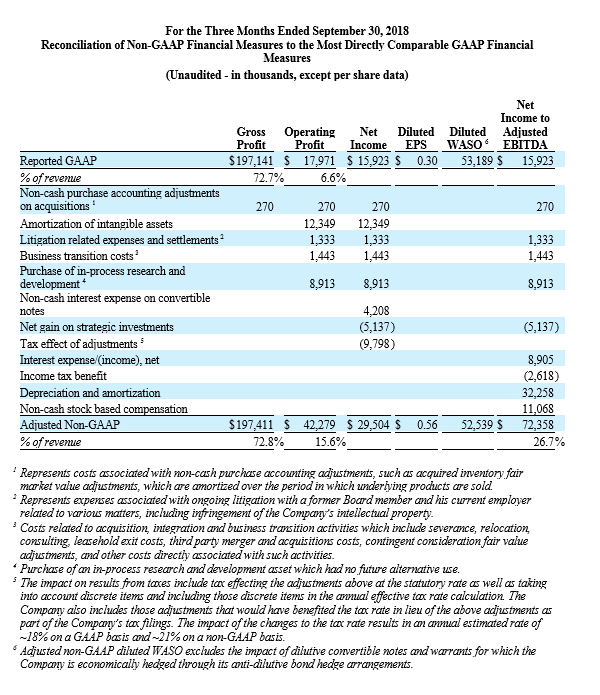
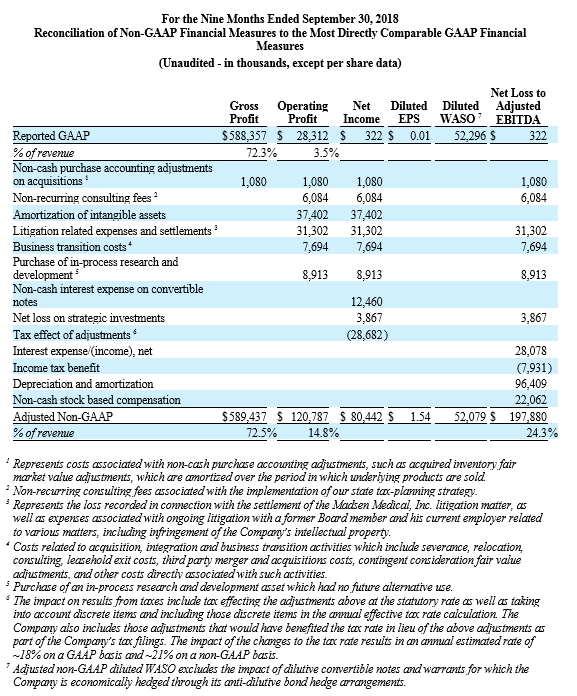
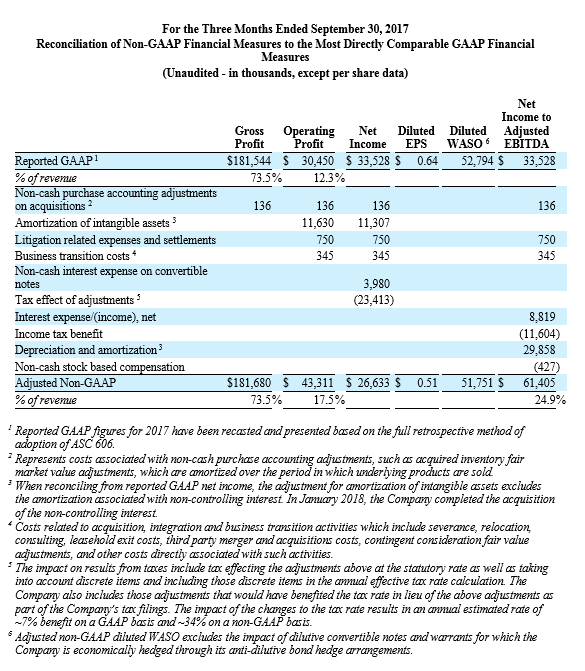
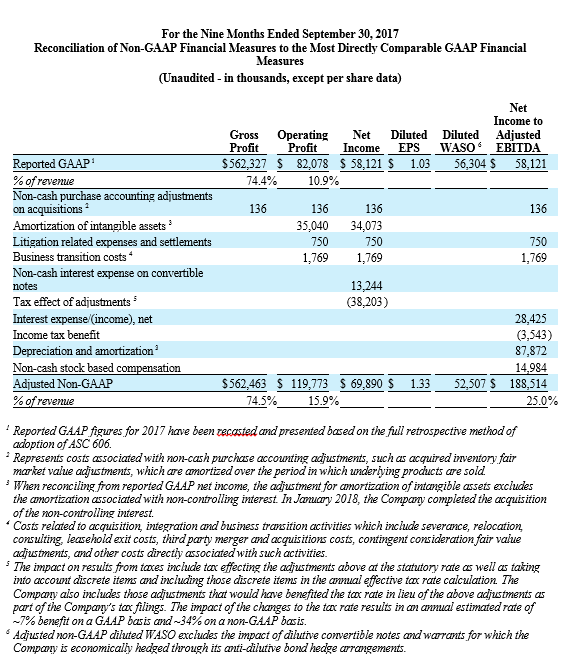
For the third quarter 2018, GAAP and non-GAAP gross profit was $197.1 million and $197.4 million, respectively, while GAAP and non-GAAP gross margin was 72.7% and 72.8%, respectively. These results compared to GAAP and non-GAAP gross profit of $181.5 million and $181.7 million, respectively, and both GAAP and non-GAAP gross margin of 73.5% for the third quarter 2017. Total GAAP and non-GAAP operating expenses were $179.2 million and $155.1 million, respectively, for the third quarter of 2018. These results compared to GAAP and non-GAAP operating expenses of $151.1 million and $138.4 million, respectively, for the third quarter 2017.
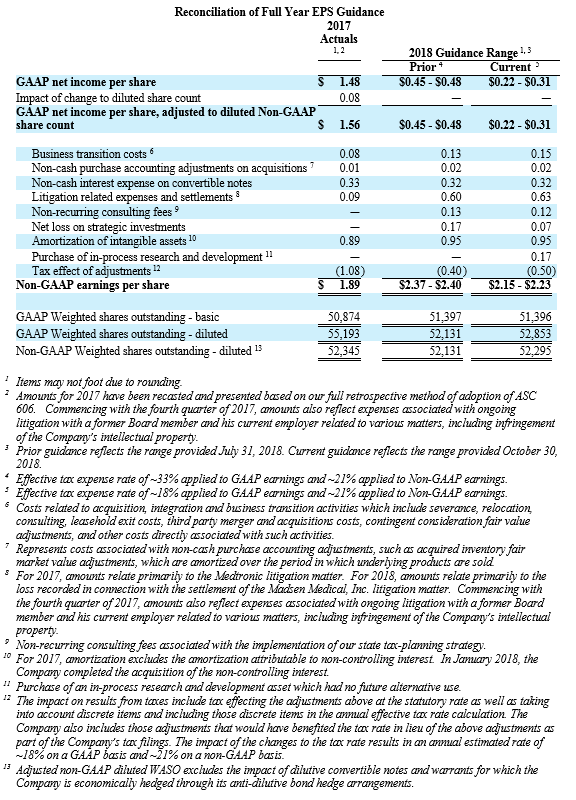
NuVasive reported GAAP net income of $15.9 million, or $0.30 per diluted share, for the third quarter 2018 compared to GAAP net income of $33.5 million, or $0.64 per diluted share, for the third quarter 2017. On a non-GAAP basis, NuVasive reported net income of $29.5 million, or $0.56 per diluted share, for the third quarter 2018 compared to net income of $26.6 million, or $0.51 per diluted share, for the third quarter 2017.
Annual Financial Guidance for 2018
The Company updated its full-year 2018 financial guidance by increasing its revenue guidance by $5 million to reflect a new range of $1,100 million to $1,110 million and reducing its non-GAAP operating margin guidance range to 15.0% – 15.5% as a result of accelerated investments in infrastructure and commercial sales force in anticipation of the overall spine market growth trending up to more historical averages.
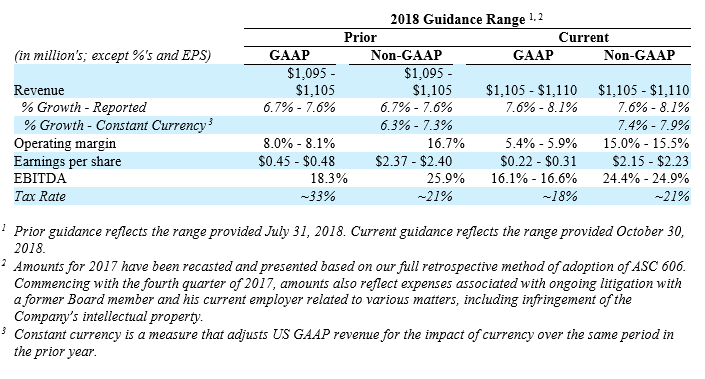
- Full-year 2018 revenue in the range of $1,105 million to $1,110 million reflecting reported growth of 7.6% to 8.1%, and growth in the range of 5.7% to 6.2%, exclusive of the SafePassage acquisition;
- Non-GAAP diluted earnings per share in a range of $2.15 to $2.23 compared with the prior expectation of $2.37 to $2.40;
- Non-GAAP operating profit margin in the range of 15.0% to 15.5%, compared with the prior expectation of 16.7%;
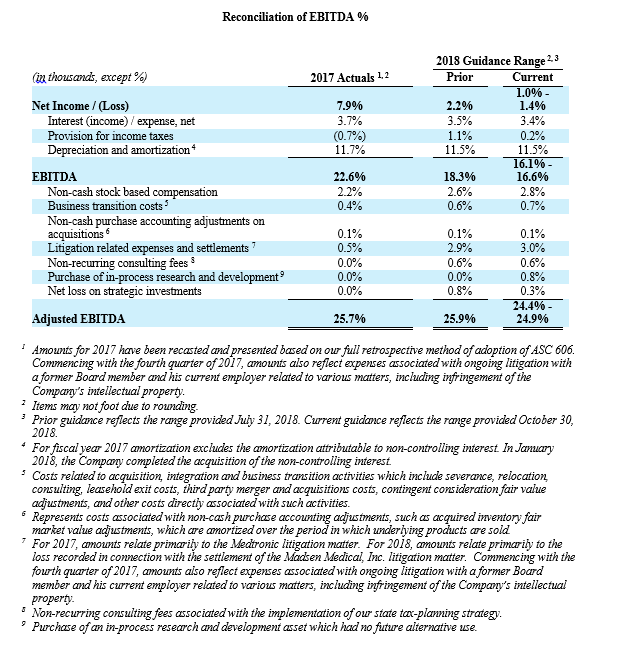
- Adjusted EBITDA margin in the range of 24.4% to 24.9%, compared with the prior expectation of 25.9%;
- Non-GAAP effective tax expense rate of approximately 21%;
- The Company expects currency to have a positive impact on revenue in 2018 of approximately $2 million compared with the prior expectation of $3 million; and
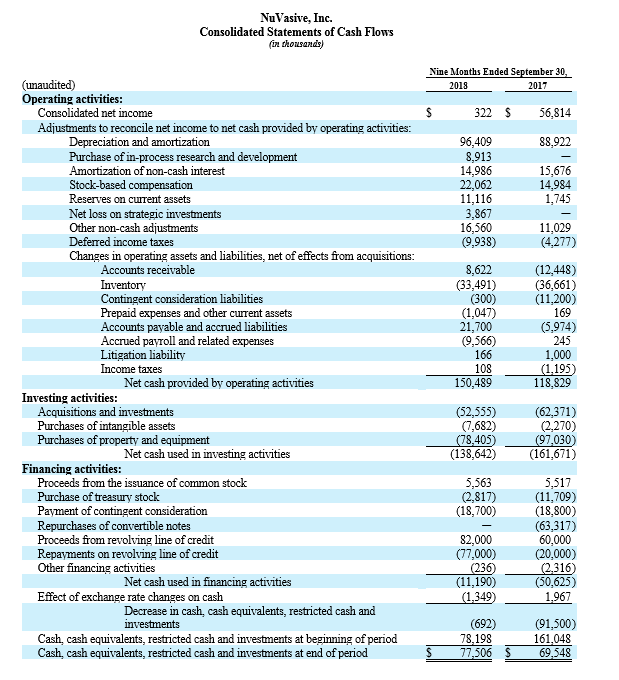
- The Company expects to drive an adjusted EBITDA of approximately $269 million to $276 million, compared with the prior expectation of approximately $283 million to $293 million.
The above guidance assumes a full-year benefit of U.S. tax reform, suspension of the medical device tax and the SafePassage acquisition.
Supplementary Financial Information
For additional financial detail, please visit the Investor Relations section of the Company’s website at www.nuvasive.com to access Supplementary Financial Information.
Reconciliation of Non-GAAP Information
Management uses certain non-GAAP financial measures such as non-GAAP earnings per share, non-GAAP net income, non-GAAP operating expenses and non-GAAP operating profit margin, which exclude amortization of intangible assets, business transition costs, purchased in-process research and development, one-time restructuring and related items in connection with acquisitions, investments and divestitures, non-recurring consulting fees, certain litigation expenses and settlements, gains and losses from strategic investments, and non-cash interest expense (excluding debt issuance cost). Management also uses certain non-GAAP measures which are intended to exclude the impact of foreign exchange currency fluctuations. The measure constant currency utilizes an exchange rate that eliminates fluctuations when calculating financial performance numbers. The Company also uses measures such as free cash flow, which represents cash flow from operations less cash used in the acquisition and disposition of capital. Additionally, the Company uses an adjusted EBITDA measure which represents earnings before interest, taxes, depreciation and amortization and excludes the impact of stock-based compensation, business transition costs, purchased in-process research and development, one-time restructuring and related items in connection with acquisitions, investments and divestitures, non-recurring consulting fees, certain litigation expenses and settlements, gains and losses on strategic investments, and other significant one-time items.
Management calculates the non-GAAP financial measures provided in this earnings release excluding these costs and uses these non-GAAP financial measures to enable it to further and more consistently analyze the period-to-period financial performance of its core business operations. Management believes that providing investors with these non-GAAP measures gives them additional information to enable them to assess, in the same way management assesses, the Company’s current and future continuing operations. These non-GAAP measures are not in accordance with, or an alternative for, GAAP, and may be different from non-GAAP measures used by other companies. Set forth below are reconciliations of the non-GAAP financial measures to the comparable GAAP financial measure.
During the quarter ended June 30, 2018, the Company began excluding from its non-GAAP financial results certain litigation related expenses associated with ongoing litigation with a former Board member and his current employer related to various matters, including infringement of the Company’s intellectual property. For consistency and comparability, the Company has re-casted non-GAAP financial results for each of the quarters ended December 31, 2017 and March 31, 2018 to exclude these litigation expenses in such periods, which were $0.4 million and $0.6 million, respectively.
Investor Conference Call
NuVasive will hold a conference call today at 4:30 p.m. ET / 1:30 p.m. PT to discuss the results of its financial performance for the third quarter 2018. The dial-in numbers are 1-877-407-9039 for domestic callers and 1-201-689-8470 for international callers. A live webcast of the conference call will be available online from the Investor Relations page of the Company’s website at www.nuvasive.com. After the live webcast, the call will remain available on NuVasive’s website through November 27, 2018. In addition, a telephone replay of the call will be available until November 6, 2018. The replay dial-in numbers are 1-844-512-2921 for domestic callers and 1-412-317-6671 for international callers. Please use pin number: 13683334.
About NuVasive
NuVasive, Inc. (NASDAQ: NUVA) is the leader in spine technology innovation, focused on transforming spine surgery and beyond with minimally disruptive, procedurally integrated solutions designed to deliver reproducible and clinically-proven surgical outcomes. The Company’s portfolio includes access instruments, implantable hardware, biologics, software systems for surgical planning, navigation and imaging solutions, magnetically adjustable implant systems for spine and orthopedics, and intraoperative monitoring service offerings. With over $1 billion in revenues, NuVasive has an approximate 2,400 person workforce in more than 40 countries serving surgeons, hospitals and patients. For more information, please visit www.nuvasive.com.
Forward-Looking Statements
NuVasive cautions you that statements included in this news release or made on the investor conference call referenced herein that are not a description of historical facts are forward-looking statements that involve risks, uncertainties, assumptions and other factors which, if they do not materialize or prove correct, could cause NuVasive’s results to differ materially from historical results or those expressed or implied by such forward looking statements. In addition, this news release contains selected financial results from the third quarter 2018, as well as projections for 2018 financial guidance and longer-term financial performance goals. The Company’s results for the third quarter 2018 are prior to the completion of review and audit procedures by the Company’s external auditors and are subject to adjustment. In addition, the Company’s projections for 2018 financial guidance and longer-term financial performance goals represent initial estimates, and are subject to the risk of being inaccurate because of the preliminary nature of the forecasts, the risk of further adjustment, or unanticipated difficulty in selling products or generating expected profitability. The potential risks and uncertainties which contribute to the uncertain nature of these statements include, among others, risks associated with acceptance of the Company’s surgical products and procedures by spine surgeons, development and acceptance of new products or product enhancements, clinical and statistical verification of the benefits achieved via the use of NuVasive’s products (including the iGA™ platform), the Company’s ability to effectually manage inventory as it continues to release new products, its ability to recruit and retain management and key personnel, and the other risks and uncertainties more fully described in the Company’s news releases and periodic filings with the Securities and Exchange Commission. NuVasive’s public filings with the Securities and Exchange Commission are available at www.sec.gov. NuVasive assumes no obligation to update any forward-looking statement to reflect events or circumstances arising after the date on which it was made.
Investor & Media Contact:
Suzanne Hatcher
NuVasive, Inc.
858-458-2240
[email protected]